UC research leader participates in COVID-19 vaccine trial
Dean for clinical research literally rolls up his sleeves to help overcome the pandemic
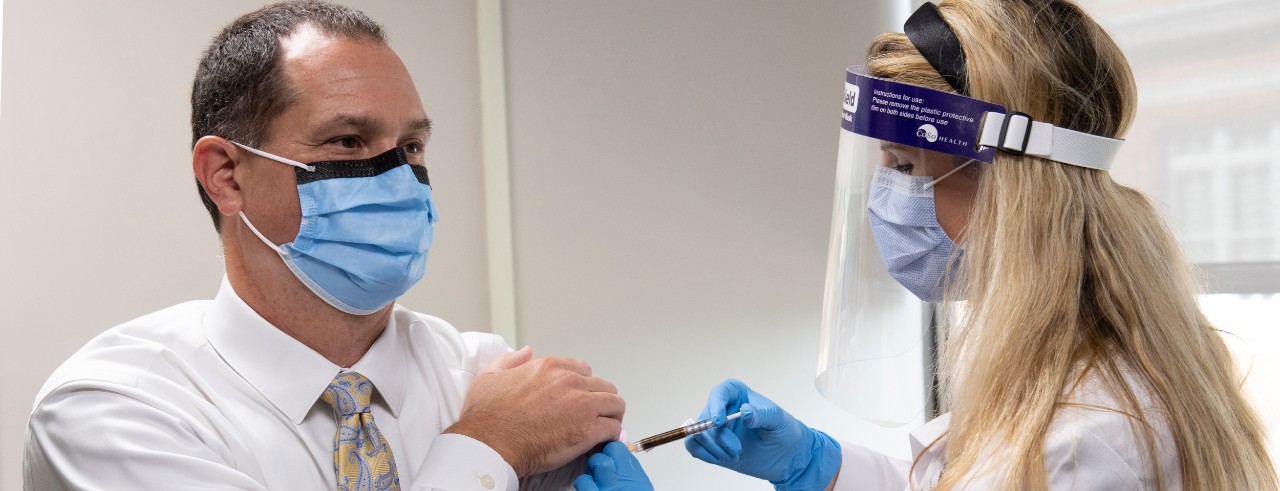
Brett Kissela, MD, pushes up the fabric on the arm of his dress shirt. He sits on an exam table on the fourth floor of Holmes Hospital, located on the University of Cincinnati’s medical campus.
“Is that enough?” he asks, as the pharmacist prepares the needle, which could contain either a vaccine for COVID-19 or a placebo. The first doses of the trial were administered last week.
“What made you interested in the trial?” asks Mary Burns, PharmD, pharmacist with UC Health, administering the dose.
“Science,” says Kissela, smiling under his mask.
The senior associate dean for clinical research at the UC College of Medicine and chief of research services at UC Health served as an example for the research he conducts, becoming a participant in the vaccine trial, sponsored by Moderna, Sept. 1. Moderna is the Massachusetts-based biotechnology company focused on drug discovery, development and vaccine technologies.
"The only way out of this is through science,” he says. “With over 20 years of experience as a researcher, this is a time when I can personally make a difference and help advance the field from the other side of the fence.”

Brett Kissela, MD, has his blood drawn as part of the clinical trial process. Photo/Colleen Kelley/UC Creative + Brand
With over 20 years of experience as a researcher, this is a time when I can personally make a difference and help advance the field from the other side of the fence.
Brett Kissela, MD

Brett Kissela, MD, participates in the COVID-19 vaccine trial at UC. He poses here with UC Health nurse Eva Whitehead. Colleen Kelley/UC Creative + Brand
Patients who participate in the blinded, randomized, placebo-controlled study will receive either the vaccine, or a placebo, in two injections. The vaccine is incorporated into the patient’s cells, which should begin producing a viral antigen. Then, the patient’s immune system can respond by making antibodies that will protect against future viral infection. Patients will keep a daily electronic diary of their symptoms, and also participate in telehealth and clinic visits with UC Health clinicians over a two-year period.
“We have to do this the right way, in order to make sure it works, and create an effective vaccine that will help our entire population,” he says. “I’m excited to be part of it.”
Featured photo of Brett Kissela, MD, by Colleen Kelley/UC Creative + Brand
Impact Lives Here
Those eligible for the COVID-19 vaccine trial include adults at least 18 years of age with no known history of SARS-CoV-2 infection, but are at risk of becoming infected. In addition to healthy individuals, patients at risk for severe COVID-19 disease, such as those with diabetes, heart or lung disease, or other chronic medical conditions, will also be included in the study.
For information or to enroll in the study, click here, call 513-245-3417, or email UCcovidresearch@uchealth.com.
Related Stories
The future, decoded. UC scholars reveal what’s next
May 20, 2026
The University of Cincinnati’s NEXT Innovation Scholars presented Gen Z-fueled insights on possible future trends at the Futures Forum 2026. Here’s what they see ahead.
6 ways starting a GLP-1 medication could affect your emotions
May 20, 2026
When patients first start taking a glucagon-like peptide-1 (GLP-1) medication, they probably expect to feel full. But they might not anticipate how it can influence their emotions. The medications act on the stomach and the brain, said Malti Vij, MD, a University of Cincinnati adjunct associate professor in the College of Medicine's Department of Internal Medicine and a diplomate of the American Board of Obesity Medicine.
UC researchers recruit older adults for extreme heat health study
May 20, 2026
The University of Cincinnati’s Center for Collaboration on Climate & Community for Health (C4H) is recruiting older adults to participate in a study tracking their health during periods of extreme summer heat.
