UC study: Subcutaneous infusion pump safe, effective for Parkinson’s treatment
Results of phase 3 trial published in the Lancet Neurology
An international, multisite phase 3 trial co-led by a University of Cincinnati researcher found Parkinson’s disease medication delivered through an infusion pump is safe and effective at reducing symptoms for longer periods of time.
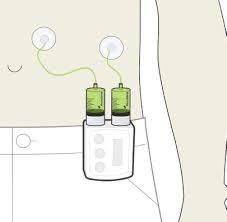
A belt pump like this can provide 24-hour, continuous levodopa to patients and was found to be safe and effective in the trial. Photo provided by NeuroDerm.
These results, published March 15 in the Lancet Neurology journal, could lead to additional treatment options for patients with the condition.
Parkinson’s symptoms such as tremors, slowness and stiffness are caused by low levels of dopamine in the body. For decades, doctors have treated Parkinson’s by giving patients levodopa, the inactive substance in the brain that once converted makes dopamine.
“Levodopa is a replacement strategy. We all make levodopa, but Parkinson's patients make less of it,” said Espay, co-principal investigator of the trial, James J. and Joan A. Gardner Family Center for Parkinson’s Disease Research Endowed Chair in UC’s Department of Neurology and Rehabilitation Medicine and a physician at the UC Gardner Neuroscience Institute.
Espay said oral levodopa is effective and typically helps people regain normal motor function, but its benefits tend to last less than a few hours after a few years, requiring increases in doses or its frequency.
Levodopa is most commonly administered orally, but this trial tested continuous, 24-hour levodopa delivery through a subcutaneous infusion pump. A total of 381 patients with Parkinson’s disease in 16 countries enrolled in the trial and were randomized to receive levodopa through the infusion pump or through traditional oral medication.
The researchers found levodopa delivered through the infusion pump was safe and led to almost two hours a day (1.72) of additional “on time,” or the time when the medication is working and symptoms are lessened, compared to taking levodopa orally.

Alberto Espay, MD. Photo/University of Cincinnati.
Espay said the results of this trial pave the way for this specific infusion pump delivery system to be approved by the Food and Drug Administration and other countries’ respective governing bodies.
“Once approved, this will become an important treatment strategy to consider for patients with Parkinson’s disease experiencing motor fluctuations not adequately controlled with medication,” he said. “Future studies will need to determine the durability of the long-term benefits and whether any safety issues could emerge, as well as how it might compare with deep brain stimulation.”
Two additional subcutaneous delivery systems are also expected to be approved this year, Espay said, and researchers are continuing to study how to improve levodopa formulations and delivery to optimize its effect for patients.
“Levodopa delivery systems are expected to continue to improve over time,” he said. “This is a thriving area of research for the benefits of our patients.”
Impact Lives Here
The University of Cincinnati is leading public urban universities into a new era of innovation and impact. Our faculty, staff and students are saving lives, changing outcomes and bending the future in our city's direction. Next Lives Here.
Featured photo at top of 3-D illustration of a dopamine molecule. Photo/Dr_Microbe/iStock.
The trial was funded by NeuroDerm.
Related Stories
CCM welcomes new film and media scoring faculty member J.R. Paredes
May 20, 2026
UC College-Conservatory of Music Dean Pete Jutras has announced the appointment of J.R. Paredes as CCM's new Assistant Professor of Film and Media Scoring. His faculty appointment officially begins on Aug. 15, 2026. Paredes is a composer, music producer and audio post-production specialist whose work spans film, television and commercial music. His credits include original scores for feature films and series distributed on platforms such as Apple TV+ and Prime Video, as well as extensive work in sound design and mixing for film and media.
6 ways starting a GLP-1 medication could affect your emotions
May 20, 2026
When patients first start taking a glucagon-like peptide-1 (GLP-1) medication, they probably expect to feel full. But they might not anticipate how it can influence their emotions. The medications act on the stomach and the brain, said Malti Vij, MD, a University of Cincinnati adjunct associate professor in the College of Medicine's Department of Internal Medicine and a diplomate of the American Board of Obesity Medicine.
UC researchers recruit older adults for extreme heat health study
May 20, 2026
The University of Cincinnati’s Center for Collaboration on Climate & Community for Health (C4H) is recruiting older adults to participate in a study tracking their health during periods of extreme summer heat.
