WLWT: A change in mammography standards could help save lives
A new Food and Drug Administration policy will soon require health care providers to tell women if they have dense breast tissue, a trait that can make cancer harder to detect. The move is aimed to support prevention, early detection and treatment of breast cancer.
The announcement also follows advocacy from patients, lawmakers and University of Cincinnati Cancer Center physicians that led to the passage of Ohio House Bill 371, which requires insurance companies to cover additional screenings for individuals who are at high risk for breast cancer or have dense breast tissue.
Erich J. Boomgarden, MD, assistant professor in UC's College of Medicine and a UC Health radiologist, told WLWT mammography is still the standard of care, but additional MRI or ultrasound screenings can help see through dense breast tissue.
"If you get your screening mammogram, and assuming it comes back negative, meaning there was nothing suspicious in there, we would recommend getting a supplemental screening in six months," Boomgarden said. "We do that because it's helpful for us because again, by staggering it that way, then anything that may be developing, between the screening mammogram and MRI hopefully will be detected sooner then."
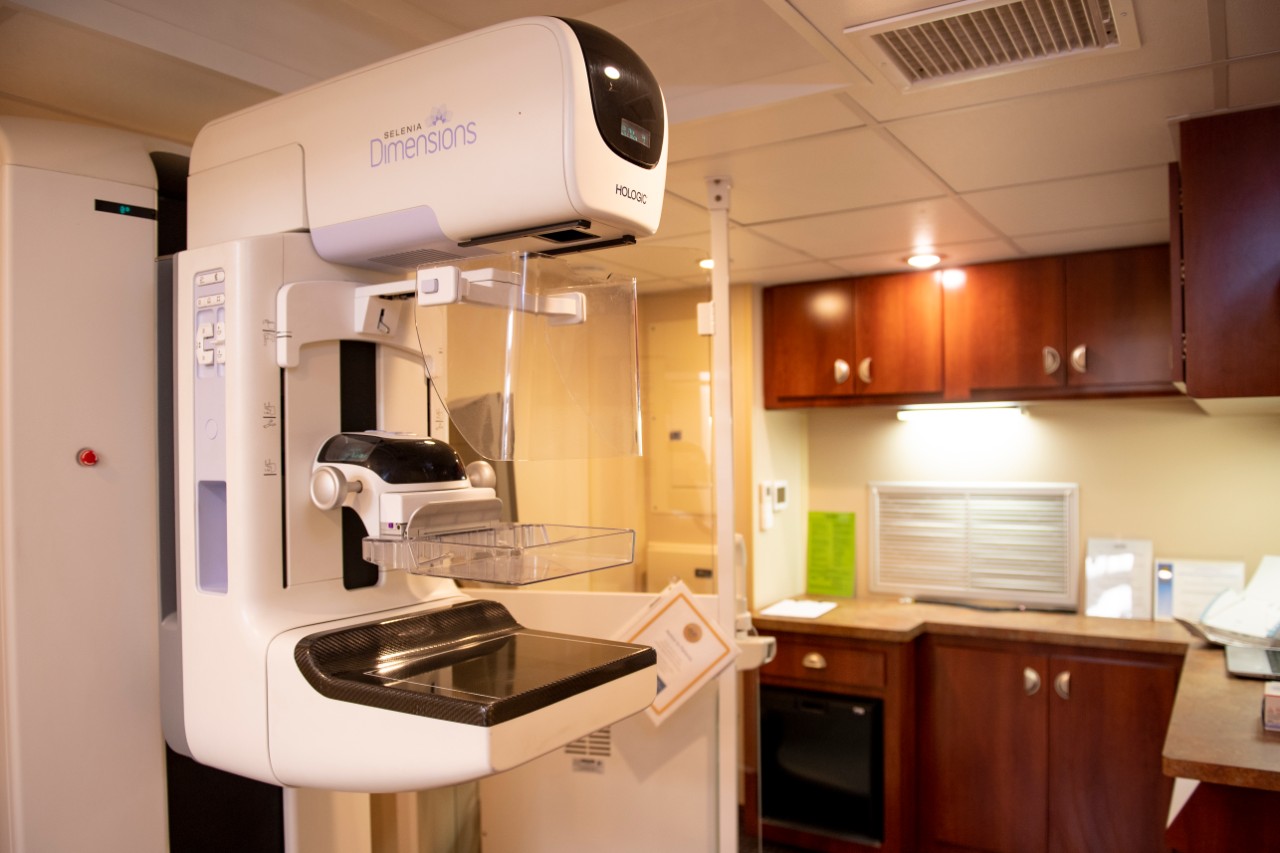
Featured photo at top of a 3D mammogram machine. Photo/Colleen Kelley/UC Marketing + Brand.
Related Stories
Three years, countless stories
May 15, 2026
UC's Klekamp Law celebrates its 193rd Hooding with stories from graduates reflecting on their paths through the college.
Driven by curiosity, guided by care
May 14, 2026
Max Wilson, a University of Cincinnati College of Allied Health Sciences health sciences major on the pre-physician assistant track, found his path expanding beyond the classroom and into hands-on research focused on human performance and patient care.
UC Blue Ash celebrates top students and recognizes Honor Student of the Year
May 14, 2026
The University of Cincinnati Blue Ash College recently hosted a special event that celebrated students for exceptional achievements during the 2025-26 academic year. The honorees included academic award winners, student engagement award winners, Latin Honors graduates, and the 2026 UC Blue Ash College Honor Student of the Year.
