WCPO: DOD-backed clinical trial begins at UC
Research aims to repair meniscus tissue, reduce the odds of surgical failure
A Department of Defense-funded clinical trial being led at the University of Cincinnati aims to repair meniscus tissue and reduce the odds of surgical procedures failing, WCPO reported.

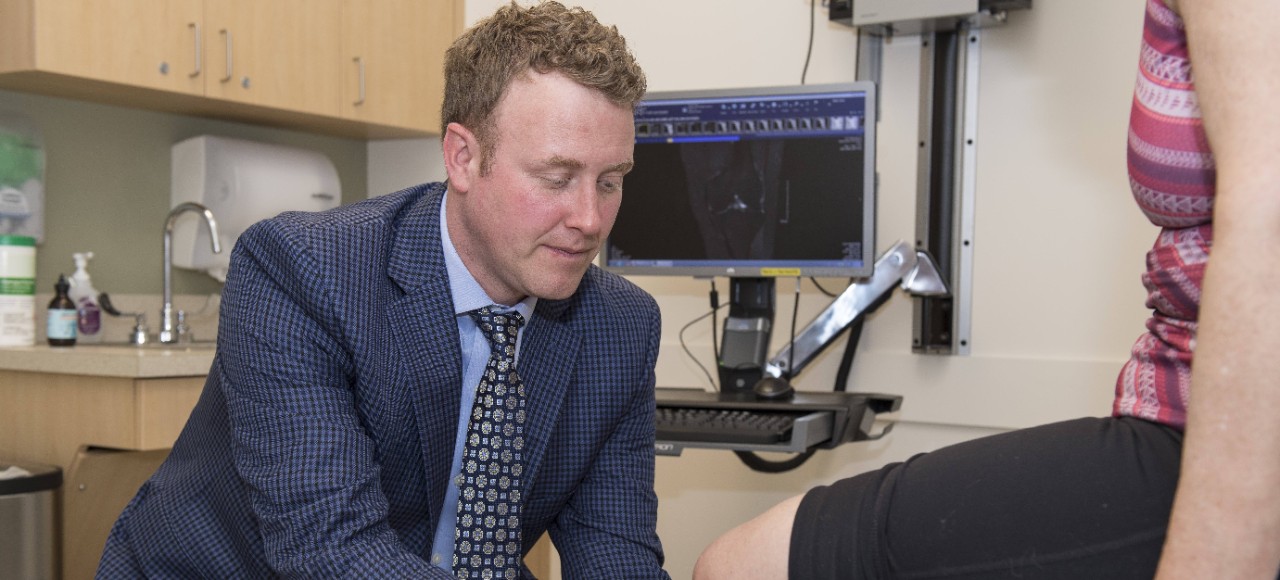
Brian Grawe
Amplicore, a UC spinoff biopharmaceutical company that is backed by the UC Venture Lab, has created a regenerative pharmaceutical solution for meniscal tears. Its injectable therapeutics are designed to be used alongside suturing of the damaged tissue to create a more pro-reparative, regenerative environment that reduces the odds of failure.
The injuries are of particular concern to the military.
“Muscular skeletal injuries in general are tremendously impactful to military personnel,” said Brian Grawe, MD, professor at the Department of Orthopedic Surgery at the UC College of Medicine. “It’s estimated that 25 million days are lost to active duty a year just on muscular skeletal injuries alone.”

James C. Y. Lin
Amplicore is using Simvastatin, an already Food and Drug Administration-approved drug used to treat high cholesterol, and redesigned its properties to create a treatment to change how meniscus tears are treated.
“The ultimate goal here is not to just treat the tissue itself but bring back the biomechanical function and for this reason, I think the military was very intrigued by our technology,” said James C. Y. Lin, PhD, Mary S. and Joseph S. Stern Jr. Professor of Orthopaedic Surgery and founder of Amplicore.
UC is the primary site for the research, and patients also will be enrolled at other sites across the United States, including Walter Reed National Military Medical Center.
Featured image at top: Brian Grawe, MD, professor at the Department of Orthopedic Surgery and the director of sports medicine in UC’s College of Medicine, is the principal investigator of Amplicore's clinical trial that seeks a regenerative pharmaceutical solution for meniscal tears.
Related Stories
Protecting the brain with chemistry
April 24, 2026
UC chemistry student Carter St. Clair will pursue his interest in computational chemistry through a new fellowship at the Air Force Research Laboratory. His topic: new applications in AI in human health.
UC, GE Aerospace celebrate Next Engineers grads
April 24, 2026
The University of Cincinnati played host in April to the graduation of this year’s class of the GE Aerospace Foundation’s Next Engineers, a global college- and career-readiness program that provides scholarship incentives for young people to become engineers.
DAAP and Peace Corps Fellows Program connects global experience to local impact
April 24, 2026
UC’s DAAP Peace Corps Fellows Program integrates global service experience with local community work, preparing students for careers in planning and development.
