
UC scientist to test anti-cocaine antibody
Antibody may help block the effects of cocaine and aid those battling addiction
Whether it is snorted, injected into the veins or smoked, cocaine delivers an intense high by increasing the levels of the natural chemical messenger dopamine that induces drug-taking behaviors, possibly leading to addiction.
But now researchers at the University of Cincinnati have developed an anti-cocaine antibody for humans that may help block the effects of cocaine and aid individuals battling drug addiction to stay in recovery.
The human antibody, reengineered in a laboratory, is now being manufactured for human trials that are expected to begin during the spring, said Andrew Norman, a professor in the UC College of Medicine’s Department of Pharmacology and Systems Physiology. When injected into the bloodstream, that antibody attaches to cocaine, preventing it from entering the brain and limiting its behavioral effects. Its success at reducing the drug’s effects have been documented in animal models of cocaine relapse.
Norman said the antibody isn’t a silver bullet, but possibly another tool in the toolbox for battling addiction.

Andrew Norman, PhD, (standing) and Terry Kirley, PhD, both professors in the Department of Pharmacology and Systems Physiology, are shown in a laboratory in the UC College of Medicine. photo/Colleen Kelley/UC Creative Services
Blunting the effects of cocaine
“It will work under a very specific set of circumstances,” said Norman. “For somebody at risk of relapse, the antibody is onboard so if they take cocaine it will be ineffective in maintaining that relapse event. It will have no effect on the brain. It won’t do anything.
“It will be pharmacologically inert, and the person will feel there is no reason to take cocaine. It will blunt the effects of cocaine,” said Norman. “The hope is by the time they take cocaine and it doesn’t do anything the cravings will dissipate.”
It will be pharmacologically inert, and the person will feel there is no reason to take cocaine. It will blunt the effects of cocaine.
Andrew Norman Professor, UC Department of Pharmacology
An earlier grant from the National Institute of Drug Abuse (NIDA) supported Norman’s findings in animal models. A recent NIDA grant of $8.4 million for a three-year period will allow Norman to test his antibody in humans, a requirement before the immunotherapy could gain approval from the U.S. Food and Drug Administration and eventual use in treating cocaine addiction.
Though cocaine use has been relatively stable since 2009, still more than 1.5 million Americans use the drug, according to the National Institute on Drug Abuse.
Norman’s work demonstrates innovation and UC’s commitment to research as described in its strategic direction Next Lives Here.
Crucial clinical trials
“This is a very important phase,” said Norman, who holds a doctorate in pharmacology. “These studies are done in healthy volunteers, not in patient populations, and just in a limited number of people. Most drugs are only in the body for a few hours, and that’s why we need to take drugs like aspirin multiple times a day. This one is different. These antibodies are expected to have a half-life — that’s the time it takes for half of it to disappear — of maybe three weeks instead of a few hours.
“People will be exposed for quite a long time, three weeks after one injection of the antibody,” said Norman.
Catalent Pharma Solutions is manufacturing the antibody under contract with UC.
Co-investigators in this study are Drs. Thomas Geracioti and Erik Nelson, professor and associate professor, respectively, in UC’s Department of Psychiatry and Behavioral Neuroscience. While the clinical trials will move forward, there is still ongoing research about the antibody that the federal FDA is requiring, said Norman. His lab is still trying to find a “maximum tolerable dose” of the antibody in animal models to guide proposed human clinical trials.
The FDA wants researchers to find a level of the antibody that would be harmful in humans by mimicking the effects in animal models. Establishing that level determines the appropriate dosage when the antibody is given as a therapy in humans. Three of four dosing levels of the antibody Norman has proposed for his clinical trial have FDA approval. Use of the fourth dosing level is pending.
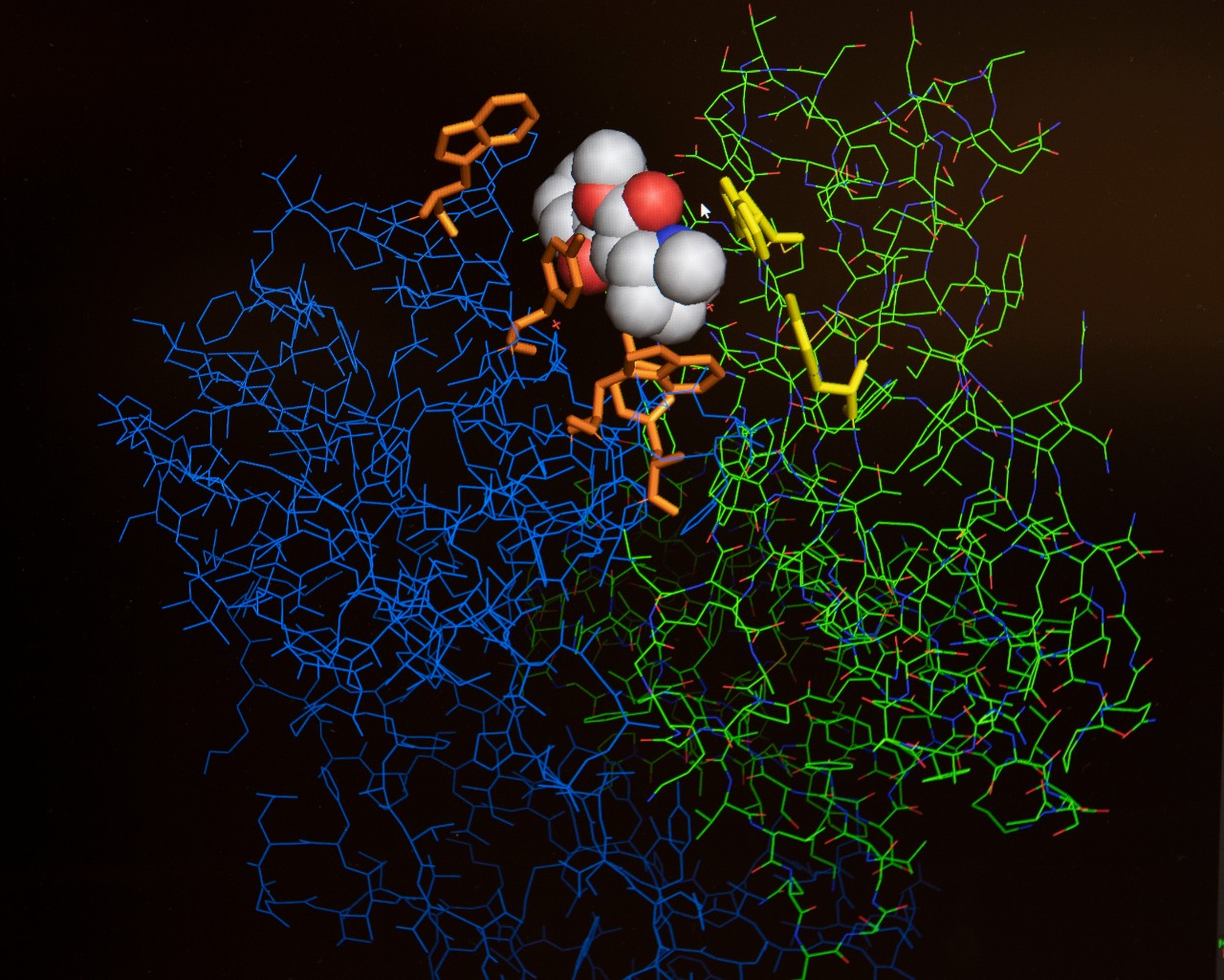
Image of the anti-cocaine antibody attached to a cocaine metabolite
“The FDA has no problem with us using those three lower doses in clinical trials,” said Norman. “But they want us to go back and try even higher doses until we find some evidence of toxicity. This won’t stop the clinical trial from preceding. The lower dosages are covered by our toxicity studies in animal models. The fourth higher dosage is on hold for now.”
Researchers say there is room for adjunct therapies, which could be psychosocial, behavioral modification or alternatively pharmacotherapy. “The antibody has no effects in the brain. It won’t block other drugs from having an effect on the body,” said Norman. “You can also apply other forms of therapy along with the antibody.”
If an individual really wants a high the antibody may blunt the effect of cocaine, but it doesn’t stop someone from trying other drugs such as opiates, methamphetamines, marijuana or alcohol, said Norman.
“These therapies are an aid to people that really want to quit and are motivated to do so” said Norman “That is true of most therapies for addictive behavior. If addicts want to get high, there is not really a lot that can be done to stop them.”

Dissected image of the human brain. Photo courtesy of Unsplash.
Next Lives Here
The University of Cincinnati is classified as a Research 1 institution by the Carnegie Commission and is ranked in the National Science Foundation's Top-35 public research universities. UC's graduate students and faculty investigate problems and innovate solutions with real-world impact. Next Lives Here.
Featured image at top shows a skeletal model of a human head. Photo/Unsplash.
As part of the disclosure, Norman said he is a co-inventor on two U.S. patents entitled "Compositions and Methods for Treating Cocaine-Related Disorders."
Related Stories
Pocket-sized population threat
June 10, 2026
The Financial Times took a deep dive into why populations around the world continue to be on the decline. The publication cited new University of Cincinnati research as part of the investigation that looks at the fall of fertility in the digital era.
Patients with developmental disabilities may benefit with an integrated care model
June 9, 2026
Researchers from the University of Cincinnati and Ohio State University have found that adults with developmental disabilities who have integrated care were less likely to go to the emergency room or be hospitalized than others who were not. Their work was published in Disability and Health Journal.
UC physician-researcher's work yields landmark five-year data for Crohn's disease drug
June 8, 2026
The University of Cincinnati College of Medicine’s Anita Afzali, MD, leads a landmark study revealing five-year efficacy and safety data for a Crohn’s disease drug called guselkumab.
