
Putting control in patients' hands
UC trial to test neurostimulation device to manage opioid withdrawal with PTSD symptoms
A new University of Cincinnati trial, in partnership with Spark Biomedical, will test a wearable neurostimulation device to help patients with opioid use disorder and post-traumatic stress disorder (PTSD) stick with medication treatment while finding the right dose.
The National Institute on Drug Abuse has awarded UC’s Joel Sprunger, PhD, a $2.1 million grant to lead the trial testing Spark Biomedical’s Sparrow Ascent device.
Study background

Joel Sprunger, PhD. Photo/University of Cincinnati.
Sprunger said in his practice at the UC Health Addiction Sciences Clinic, he often sees patients with both opioid use disorder (OUD) and PTSD.
“The rates of substance use disorder among people that have PTSD is relatively higher than other psychological disorders, and these happen to be co-occurring concerns that feed off of one another, making them difficult to treat,” said Sprunger, assistant professor of psychiatry and behavioral neuroscience in UC’s College of Medicine. “The needs of someone struggling with both PTSD and OUD can be a lot greater than if someone were struggling with one or the other. When someone is looking to make a change through treatment, it takes a lot of trust for people to engage in a therapeutic relationship, especially if they’ve been failed by trusted others before.”
For both opioid withdrawal and PTSD, Sprunger said symptoms of these syndromes often affect the body’s sympathetic nervous system’s in ways relevant to the fight-or-flight response.
“With opioid withdrawal symptoms, you can experience things like anxiety, irritability, and sweating and people feel on edge and activated,” he said. “With PTSD, there’s overlap in terms of anxiety and irritability, but also a lot of hypervigilance, exaggerated startle responses, problems sleeping, and concentration difficulties.”
The medication buprenorphine is an effective treatment to help patients with OUD manage symptoms of opioid use disorder withdrawal, but there is an adjustment period of up to three months as each patient finds the right dose to manage their symptoms.

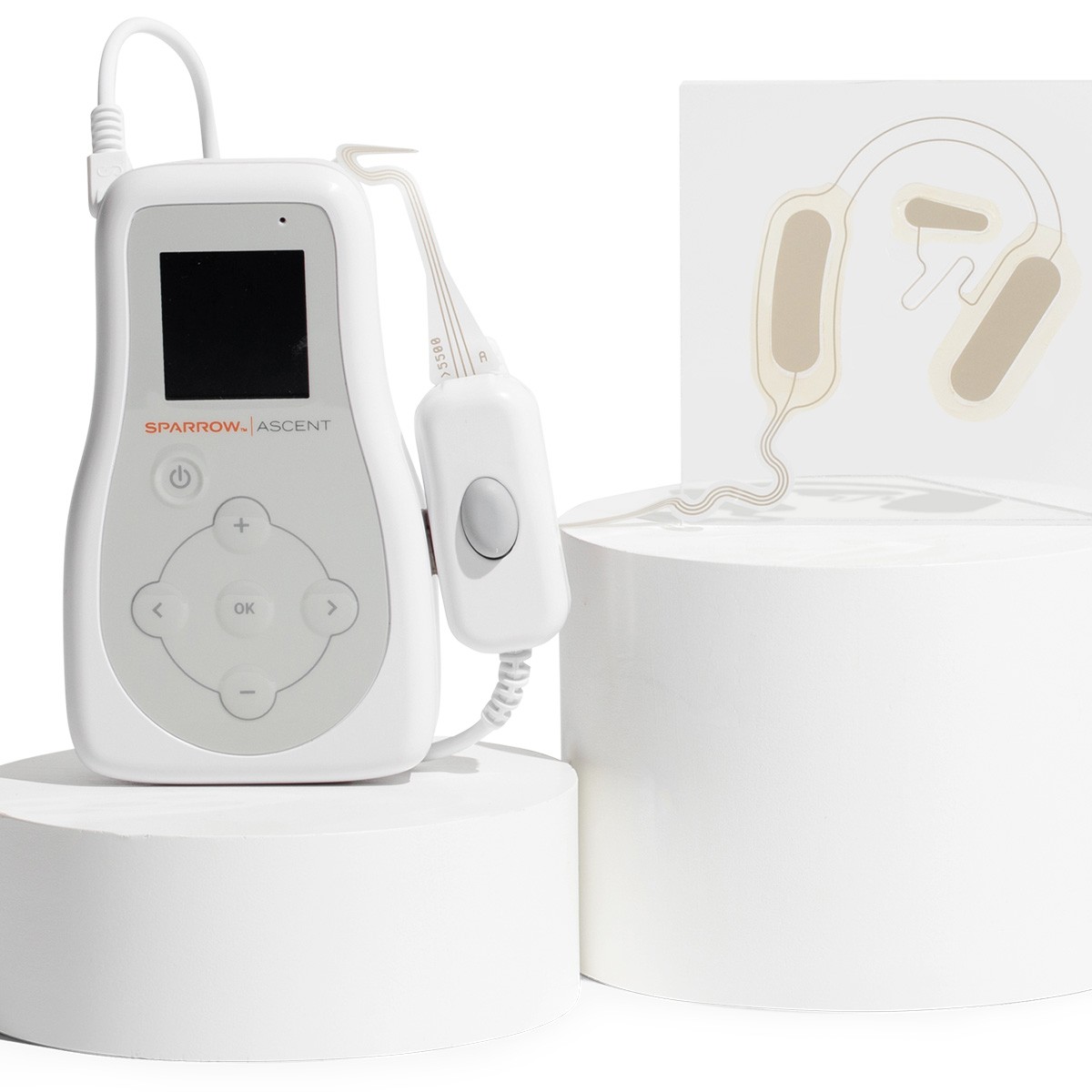
The Sparrow Ascent device is worn around the ear and gives patients a tool that gives them direct control to help manage OUD and PTSD symptoms. Photo provided by Spark Biomedical.
About 50% of people drop out of buprenorphine treatment during the dose adjustment period, and patients with PTSD are at even higher risk for stopping treatment.
In response to this challenge, the trial will utilize the Sparrow Ascent – a patient-administered wearable device that delivers mild electrical stimulation to the cranial branches of the vagus and trigeminal nerves on and around the ear.
Sprunger said the stimulation “pumps the brakes” on the sympathetic nervous system by enhancing parasympathetic activity, helping to transition someone from fight-or-flight to “rest and digest.”
This method, known as transcutaneous auricular neurostimulation (tAN), is FDA-cleared for relieving opioid withdrawal symptoms and may also aid in alleviating symptoms of PTSD. The trial will test the safety and effectiveness of tAN in patients beginning buprenorphine therapy with these co-occurring conditions.
“By providing people control over that stimulation, we can empower them with a way to turn it on when needed and feel some relief,” he said. “Their heart rate slows down, breathing slows down and there’s a lot less of a sense of panic and urgency. So that’s the key ingredient that we think will hit both PTSD and opioid withdrawal symptoms at the same time.”
The Sparrow Ascent device is FDA-cleared for relieving opioid withdrawal symptoms and may also aid in alleviating symptoms of PTSD. Photo/Spark Biomedical.
Worn on and around the ear, the device is connected by a cable to a controller. Users can activate the stimulation whenever they need it and control its intensity. It can be worn up to 24 hours a day, and the earpiece is disposable and can be replaced each day.
“Autonomic imbalance is a common occurrence in PTSD, which is characterized by experiencing high sympathetic drive (‘fight or flight’) and low parasympathetic drive (‘rest and digest’),” said Navid Khodaparast, PhD, chief science officer at Spark Biomedical. “Additionally, PTSD sufferers experience lower levels of endorphin production which can lead to emotional distress. In this study, we believe tAN therapy will improve these PTSD symptoms, and ultimately help maintain long-term BUP treatment.”
A total of 20 patients at the Gibson Center for Behavioral Change in Cape Girardeau, Missouri with opioid use disorder and PTSD taking buprenorphine as part of their standard of care will be randomized to either receive the stimulation through the Sparrow Ascent or active sham stimulation at a level that won’t activate the targeted nerves. While there is a minimum required threshold of use each week of the 12-week study, Sprunger said patients can use the device as much as they feel is needed to help manage their symptoms.
In addition to its effect on physical symptoms, Sprunger said the device also gives represents a tool for patients that provides some direct control over a part of their recovery.
“I’m a psychologist, and I see my patients for up to an hour once a week. We can do some meaningful work in that hour, but there are many hours between then and when I might not see that person again,” he said. “It’s really important that people have something they are in control of that can provide some relief whenever they need the support. Being able to put something in their hands that they can use to feel better, especially one that is sensibly a healthy coping strategy, is also very powerful.”
Impact Lives Here
The University of Cincinnati is leading public urban universities into a new era of innovation and impact. Our faculty, staff and students are saving lives, changing outcomes and bending the future in our city's direction. Next Lives Here.
Featured photo at top of woman wearing the Sparrow Ascent device. Photo/Spark Biomedical.
Related Stories
CCM welcomes new film and media scoring faculty member J.R. Paredes
May 20, 2026
UC College-Conservatory of Music Dean Pete Jutras has announced the appointment of J.R. Paredes as CCM's new Assistant Professor of Film and Media Scoring. His faculty appointment officially begins on Aug. 15, 2026. Paredes is a composer, music producer and audio post-production specialist whose work spans film, television and commercial music. His credits include original scores for feature films and series distributed on platforms such as Apple TV+ and Prime Video, as well as extensive work in sound design and mixing for film and media.
6 ways starting a GLP-1 medication could affect your emotions
May 20, 2026
When patients first start taking a glucagon-like peptide-1 (GLP-1) medication, they probably expect to feel full. But they might not anticipate how it can influence their emotions. The medications act on the stomach and the brain, said Malti Vij, MD, a University of Cincinnati adjunct associate professor in the College of Medicine's Department of Internal Medicine and a diplomate of the American Board of Obesity Medicine.
